Prescription Opioids Directory
Clinical guides and safety protocols for DEA-regulated opioid medications in the United States.
Opioids (also known as narcotics) are powerful analgesics that interact with opioid receptors in the brain to reduce the perception of pain. In the United States, these are classified as controlled substances due to their potential for dependence and overdose. Check our Opioid Safety Hub for overdose prevention protocols.
This clinical directory covers the pharmacology, safety protocols, and regulatory requirements (DEA) for the most common opioids prescribed in modern American medicine.
Explore Opioids

Oxycodone
Potent Schedule II opioid for severe acute and chronic pain management.

Fentanyl
Highly potent synthetic opioid available in patches, vials, and lozenges.

Hydrocodone
Commonly prescribed Schedule II opioid, often combined with acetaminophen.

Morphine
The gold standard for moderate-to-severe pain; derived from the opium poppy.
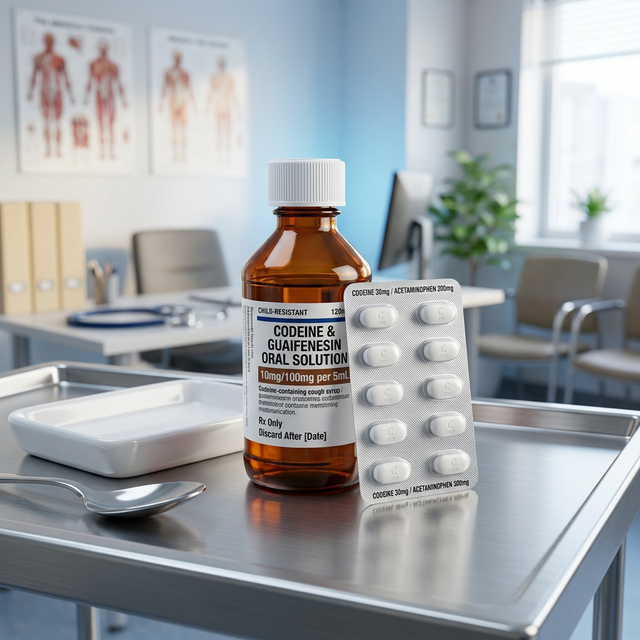
Codeine
Mild-to-moderate opioid used for pain and as an antitussive (cough suppressant).
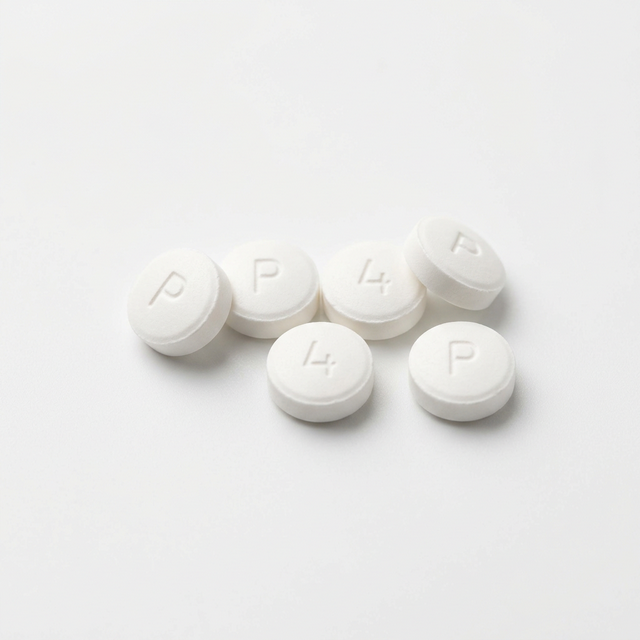
Hydromorphone
Strong analgesic (Dilaudid) used for severe pain unresponsive to other opioids.

Methadone
Long-acting opioid used for pain management and medication-assisted treatment (MAT).
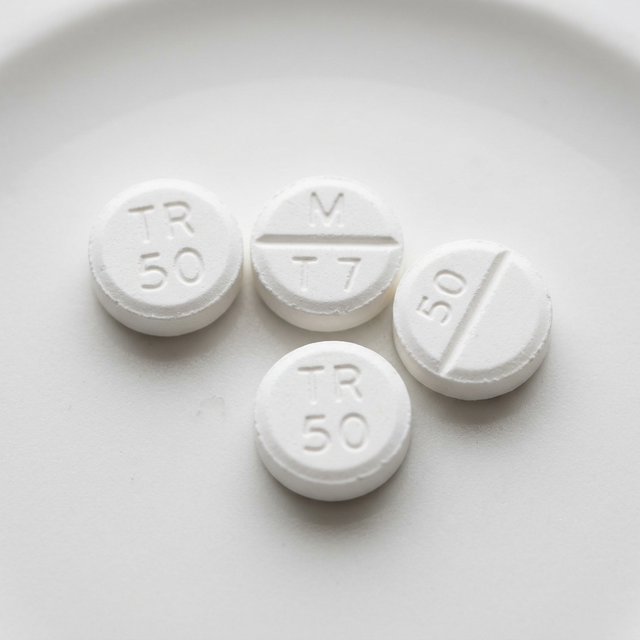
Tramadol
Schedule IV synthetic opioid with dual-action properties (opioid and SNRI).

Tapentadol
Modern Schedule II opioid (Nucynta) for severe neuropathic and acute pain.
Common Questions & Safety
What is a 'Schedule II' controlled substance?
In the United States, Schedule II drugs are substances with a high potential for abuse, but which also have a currently accepted medical use. Their distribution is strictly monitored by the DEA.
What is MME (Morphine Milligram Equivalents)?
MME is a standard clinical unit in the U.S. used to compare the potency of different opioids to ensure safe prescribing limits.
Are all opioids synthetic?
No. Natural opioids (Opiates) come from the poppy plant, semi-synthetic ones are modified from natural sources, and fully synthetic ones are lab-made.